 This change indicated that there were small errors in the previously accepted atomic weights of several of the elements and large errors for several others, for which wrong multiples of the combining weights had been used as atomic weights (the combining weight being that weight of an element that combines with a given weight of a standard). The great value of the periodic law was made evident by Mendeleyev’s success in 1871 in finding that the properties of 17 elements could be correlated with those of other elements by moving the 17 to new positions from those indicated by their atomic weights. Predictive value of the periodic law Discovery of new elements By the mid-20th century this version of the table had become the most commonly used. This class of table, too, can be greatly simplified by removing the lanthanoid and actinoid elements to a separate area. The multiple tie lines connecting the periods in the Bayley-type table are thus dispensed with. Werner in 1905, divides each of the shorter periods into two parts, one at either end of the table over the elements in the longer periods that they most resemble. Other versions of the periodic tableĪlternate long forms of the periodic table have been proposed. A useful compromise is to compress the period of 32 elements into 18 spaces by listing the 14 lanthanoids (also called lanthanides) and the 14 actinoids (also called actinides) in a special double row below the other periods. The principal disadvantage of this table is the large space required by the period of 32 elements and the difficulty of tracing a sequence of closely similar elements. The elements in each period may be connected by tie lines with one or more elements in the following period. In this table there are periods of increasing length between the noble gases the table thus contains a period of 2 elements, two of 8 elements, two of 18 elements, one of 32 elements, and an incomplete period. This was interpreted in terms of the electronic structure of atoms by Niels Bohr in 1922. The “short-period” form of the periodic table, with Groups 0, I, II,…VIII, became popular and remained in general use until about 1930.īased on an earlier (1882) model of T. With the discovery of the noble gases helium, neon, argon, krypton, radon, and xenon by Lord Rayleigh (John William Strutt) and Sir William Ramsay in 1894 and the following years, Mendeleyev and others proposed that a new “zero” group to accommodate them be added to the periodic table. Short-period form of periodic system of elements, 1930 SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find. 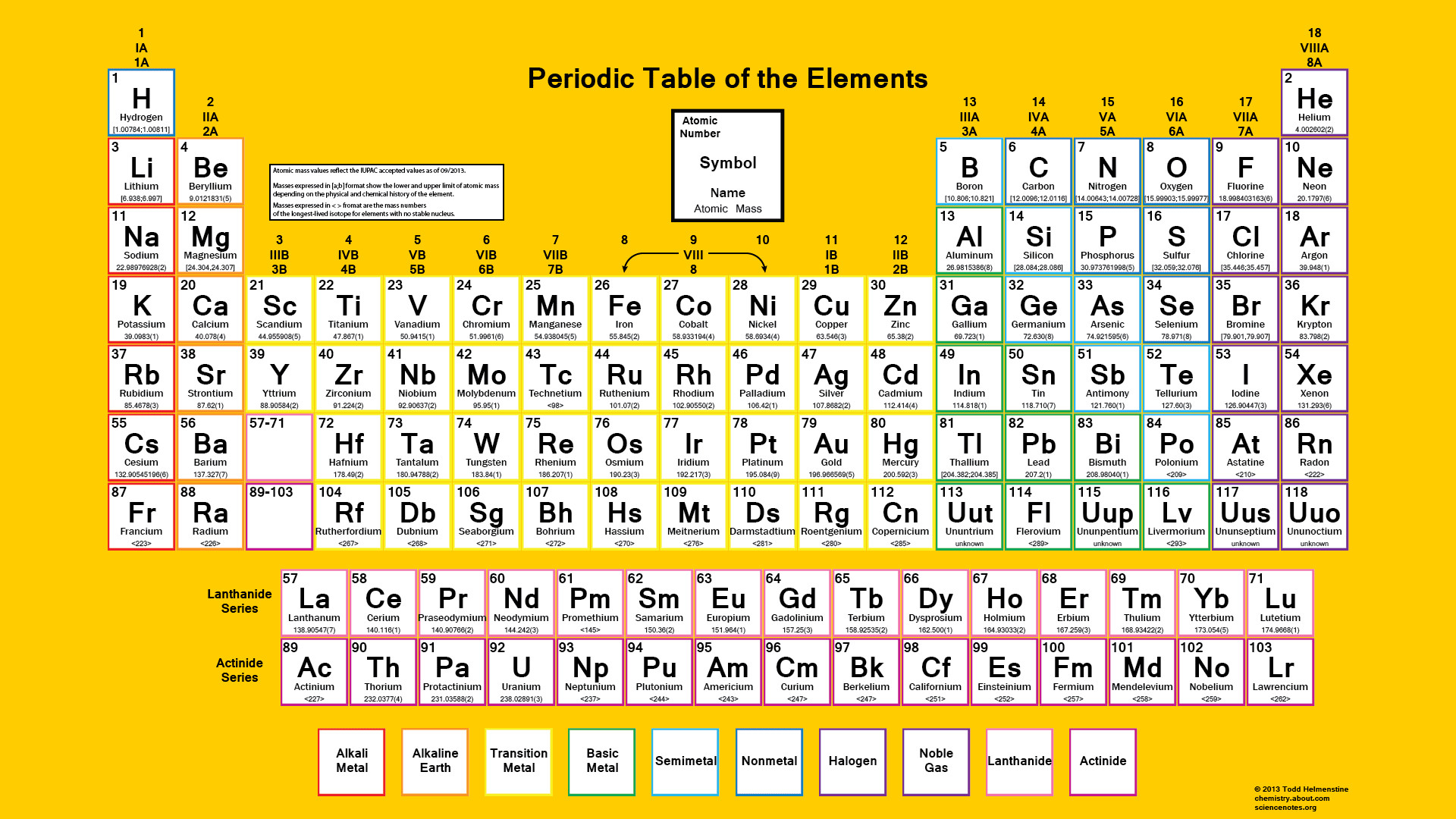
Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
